FDA Approves Zeiss' Less Invasive Laser System For Nearsightedness
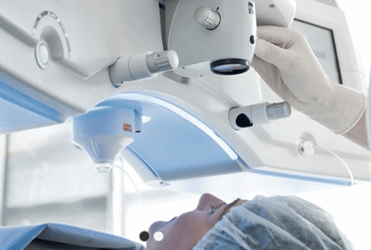
The FDA has approved the VisuMax Femtosecond Laser for small incision lenticule extraction (SMILE) procedures, which are indicated for the correction of nearsightedness in patients age 22 and older. Carl Zeiss Meditec (Zeiss) developed the SMILE procedure as a less-invasive alternative to LASIK and PRK surgery.
LASIK, one of the first laser eye corrections developed, uses one laser to create a flap in the cornea and a separate laser to reshape the cornea and correct vision. In comparison, the SMILE procedure, which is the third generation of laser eye surgery, uses an all-in-one laser system to cut a slim disc of tissue (lenticule) which is then removed through a tiny incision. The removal of tissue causes the shape of the cornea to permanently change, which corrects nearsightedness. Authors of a 2012 comparison study argued that the simpler, less invasive procedure could reduce risk of side effects and procedure costs.
“With SMILE, ophthalmic surgeons now for the first time have a refractive surgery procedure that provides great benefits for their patients with the least possible intervention; the cornea of the eye remains intact as much as possible, the predictability of correction is very good,” said Ludwin Monz, president and CEO of Zeiss, in a 2014 press release announcing the expansion of a US clinical trial investigating the procedure.
Since the technology was introduced in 2011, the SMILE procedure has been performed on over 500,000 patients in 61 countries worldwide, including Europe, China, Australia, Canada and India. To secure premarket approval (PMA) from the FDA, Zeiss submitted clinical data from five investigational sites in the US that evaluated the procedure in 328 participants over six months. All patients but one achieved visual acuity of 20/40 or better, and 88 percent had uncorrected vision of 20/20.
“This approval expands the surgical treatment options available to patients for correcting nearsightedness,” said Malvina Eydelman, director of Opthalmic and Ear, Nose, and Throat Devices in the FDA’s Center for Devices and Radiological Health (CDRH).
Monz remarked in a recent press release that the approval of SMILE with the VisuMax Laser System marks the start of “a new era” in ophthalmology for Zeiss, and that the milestone “underscored Zeiss’ commitment” to innovative technology.
According to Jim Mazzo, recently appointed global president of ophthalmic devices at Zeiss, the company will continue expanding clinical indications for the SMILE procedure. Mazzo added that Zeiss currently is conducting a clinical trial, investigating the system in patients with myopia with astigmatism, which is expected to be complete in March of 2017.
